
EUROSPINE 2025, here we come, waiting for you at booth #FG20
Welcome To The Eurospine 2025
Our booth number: 7.2b Budapest #FG20
October 22–24, 2025
Bella Center, Copenhagen, Denmark
Official exhibition website: https://www.eurospine.org/

Welcome To The Eurospine 2025
Our booth number: 7.2b Budapest #FG20
October 22–24, 2025
Bella Center, Copenhagen, Denmark
Official exhibition website: https://www.eurospine.org/

Welcome To The North American Spine Society (NASS 2025)
Our booth number: #851
November 14-16, 2025
Colorado Convention Center Denver, CO, USA
Official exhibition website: https://www.spine.org/AM

Welcome To The German Congress For Orthopedics And Trauma Surgery(DKOU 2025)
Our booth number: #B38
CityCube Berlin, Germany October 28–31, 2025
Official exhibition website: https://dkou.org/en/

We are delighted to announce that CHANGMEI's kyphoplasty system products have successfully obtained FDA approval. This significant milestone includes our kyphoplasty balloon catheter, kyphoplasty tool kit, and balloon inflator.
This approval is a testament to our commitment to providing high-quality medical devices that meet stringent regulatory standards. It reflects our dedication to innovation and excellence in the field of orthopedic surgery.
We are excited about the opportunities this approval brings and look forward to offering our enhanced product line to healthcare professionals and their patients.
Thank you for your continued support and confidence in CHANGMEI.
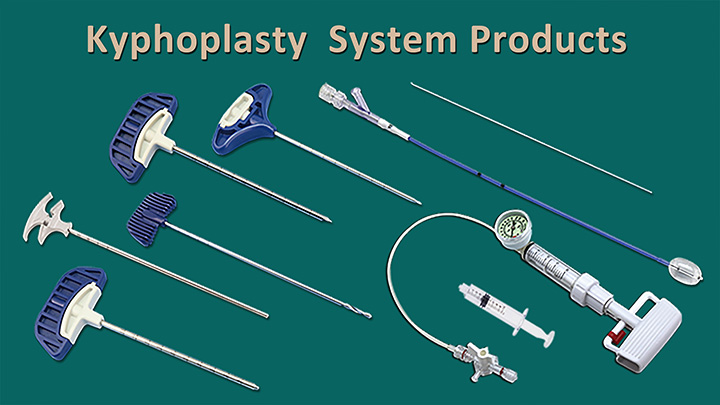
Jiangsu Changmei Medtech Co., Ltd.'s achievement in obtaining the EU MDR CE certificate for its Kyphoplasty System Products(kyphoplasty balloon catheter, kyphoplasty tool kit, and balloon inflator) represents a watershed moment in the company's journey toward global recognition and success.

Scan the QR code to download Changmei product brochure, Kyphoplasty System Products and Endoscopic Products Brochure.

The sun is just sailing in, and the dream is built together. On the morning of July 3, the groundbreaking ceremony of the West Taihu Plant of Jiangsu Changmei Medtech Co., Ltd. [WinMed Ultra-high Pressure Balloon R&D and Industrialization Project] was successfully held. Changmei medical management personnel, construction unit, design unit, project management company leaders participated in this event. Yuan Luxin, general manager of Jiangsu Changmei Medtech Co., Ltd., Fang Peiqi, chairman of Jiangsu Tianqi Construction Co., Ltd., Liu Kang, general manager of Jiangsu Yingte Engineering Consulting Design Management Co., Ltd., and Wang Junjie, general manager of Jiangsu Baoji Construction Project Management Co., Ltd., all came to the stage to deliver speeches. He expressed his congratulations and expressed his willingness to cooperate with each other, do a good job in detail, and welcome the perfect completion of Jiangsu WinMed Medical's ultra-high pressure balloon research and development and industrialization project in May 2023.

Before Chinese new year, we have attended two global medical exhibitions, Arab Health in Dubai and MD&M west in USA, now our following exhibition plan is for your reference, welcome to visit our booth at your convenience, you will find new cooperation partner there.
1.ICMD Shanghai China May 14 to 17 Booth number: 6.1 U55
2.HOSPITALAR Sao Paulo Brazil May 21 to 24 Booth number: 17-166
3.MEDTEC Shanghai China Sep.25 to 27 Booth number: E401
4.EUROSPINE Helsinki Finland Oct.16 to 18 Booth number: 20B
5.COMPAMED Duesseldorf Germany Nov.18 to 21 Booth number: 8BN35-B

Changmei Medtech will attend the MD&M 2019 at Anaheim Convention Center Anaheim, CA, USA. Welcome to visit us.
MD&M 2019 5-7 February 2019 Booth number: 945

Changmei Medtech will attend the Medica 2018 at Dusseldorf Germany and Arab Health 2019 at Dubai UAE, welcome to visit us.
1. Medica 2018 12-15 November 2018 Booth number: Hall 6 D48 E1
2. Arab Health 2019 28-31 January 2019 Booth number: Hall Z4 A50